|
It is white solid and has a very high melting point. Binary compoundsĪre formed by the combination of beryllium with non-metals such as Beryllium Beryllium has a +2 oxidation allowsįorming many covalent dissolves in non- oxidizing acids. While bonding with the other atoms, it has a very high ionization In the air, beryllium resists oxidation and is extracted by the These characteristics make unique stability under thermal loading. Very high melting point which makes beryllium metal the best heat dissipationĬharacteristics per unit has a low coefficient of linear thermalĮxpansion. It has a close-packed hexagonal crystal has a Emerald is known to be a variety of beryl. Friedrich Wohler at berlin in 1828 isolated the beryllium metal.īeryllium generally occurs in the earth’s crust to the extent of 0.0002 Beryllium was discovered inġ798 by French chemist Nicolas-Louis Vauquelin. Of alkali metals are often insoluble in water ALKALINE METALSġ metals are less reactive than group 2 metals. Group 2 elements are called alkaline metals because they formĪlkaline solutions, hydroxides, when reacting with water and their oxides areįound in the earth’s crust. GROUP 2 OF THE PERIODIC TABLE Group 2 elements are: Br To show patterns in atomic radius, ionization energy, and electronegativity. Same group elements have similar properties and reactivity. Each group of elements having the same number of valence electrons. S-, p- and d-block elements of the periodic table are arranged into these columns The 18 vertical columns of the table are called Groups. There are also more specific groups like alkali metals, transition metals, rare metals, alkaline earth, halogens, and noble gases. There are multiple ways of grouping the elements but they are commonly divided into metals, non-metals, metalloids. In the periodic table of chemical elements, there is a group (also known as a family) is a column of elements. Side of the periodic table GROUPING METHOD 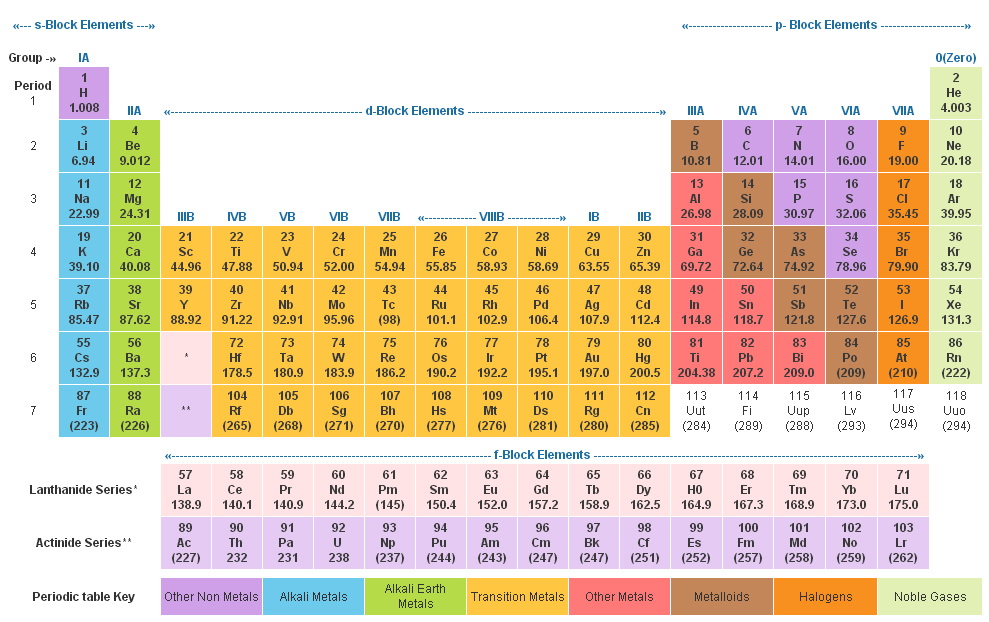
Metals are on the left side and non-metal are on the right Horizontally in order of increasing atomic number and vertically according to In the periodic table, chemical elements are present in rows The seven rows of the table are called a periodic table. The unreactive nature of the group 18 elements is due to their inability to gain or lose electrons due to their full outer shell.It is a tabular display of chemical elements, arranged in order of atomic number in rows so that elements with similar atomic structure and recurring chemical properties appear in vertical columns. All the gases in group 18 are similar in properties being heavy, unreactive and having full outer shells. This is in contrast the elements in Group 18 (Noble Gases) are relatively unreactive and are all gases. 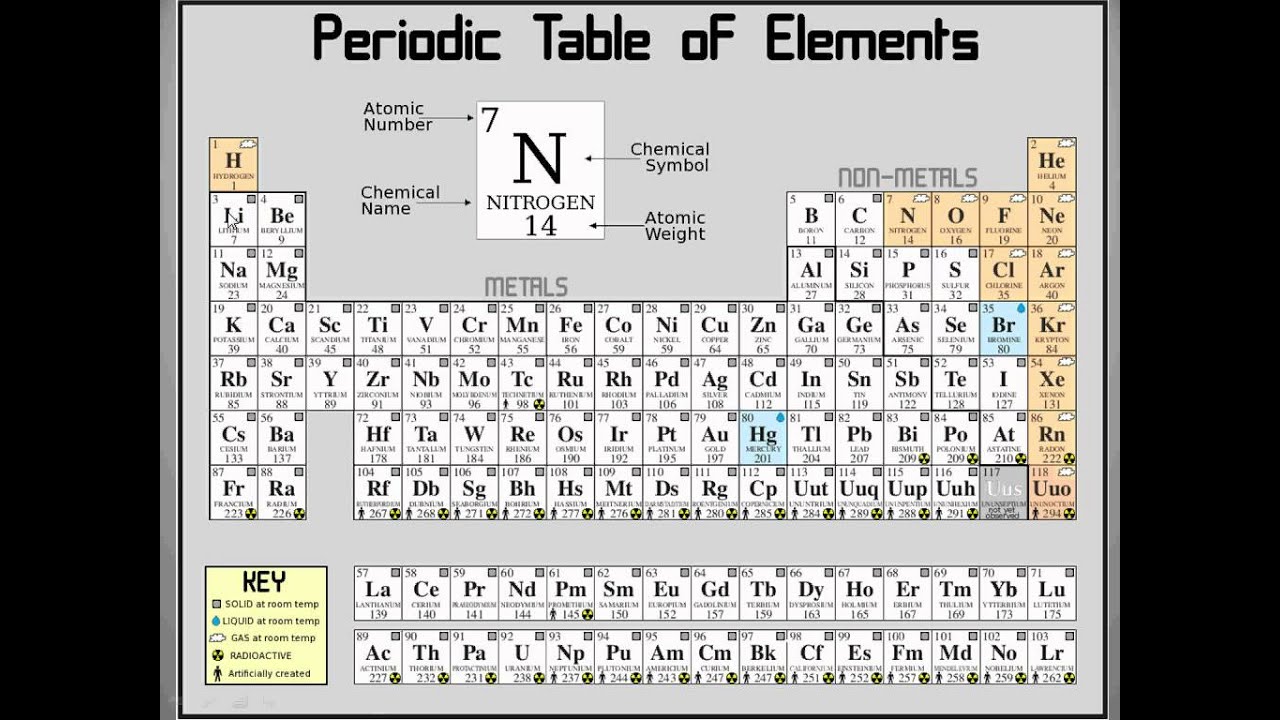 /the-periodic-table--digital-illustration--73016803-598b218ec41244001024af78.jpg)
It is these similarities in reactivity that have allowed scientists to group chemicals together into groups based on their chemical and physical similarities. /accurate-illustration-of-the-periodic-table-82020791-57cc76f23df78c71b66efbd7.jpg)
Lithium will react with water giving a small fizz and bubbling but when reacting caesium with water it reacts violently exploding and creating a lot of heat. this can be seen in the observations of the reaction. All group 1 metals will react with water to produce a hydroxide solution and hydrogen gas but progressing down the group 1 the reaction gets more violent as the outer electron (valence electron) is lost a lot more easily. There are 18 groups in the periodic table spanning from left to right.Īn example of this is the reaction of Group 1 metals with water. Scientists constructing groups of the periodic table were able to study how elements reacted with other elements and spot common similarities and differences. When referring to reactivity of an element this is its relative ease of creating a compound when reacting with another element to form a compound, this often relates to how easy that element will lose or gain electrons. The groups are the vertical columns of the periodic table. The modern periodic table is not only constructed on the increasing atomic number but also using the work of Newlands, elements are divided into groups of similar physical properties and reactivities.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
